The first study of NMN in humans was just published.
https://www.jstage.jst.go.jp/…/advpub_EJ19-03…/_pdf/-char/en
This Japanese study looked at a single dose of 100, 250, or 500 mg NMN capsules to 10 men 40-60 years old.
No adverse effects were noted.
The single oral administration of NMN was safe and effectively metabolized in healthy men without causing any significant deleterious effects. Thus, the oral administration of NMN was found to be feasible, implicating a potentialtherapeutic strategy to mitigate aging-related disorders in humans.
They did measure MeNAM 2Py and 4Py which are end products when NAD+ is utilized. 2py and 4py, show a very nice steady increase over 300 minutes after dosage.

Very fast appearance in bloodstream
One thing that jumps out from these charts is that both metabolites begin increasing at the 60 minute mark, and show steady increase over the 5 hours depicted.
This fast uptake and utilization indicates that some NMN seems to be bypassing the GI tract and liver.
Different pathway than oral NR supplements
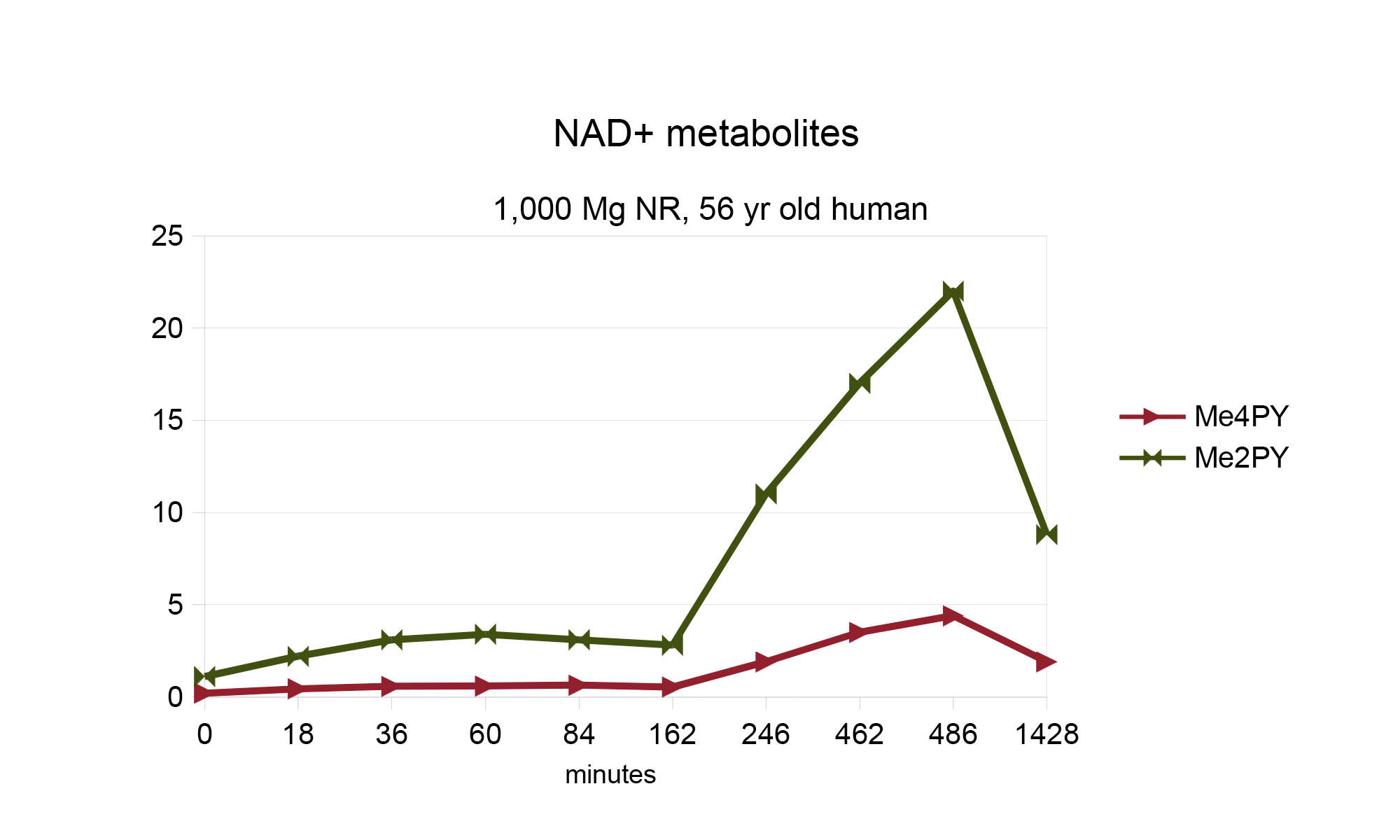
The speedy uptake is in stark contrast with data from the first human study of Nicotinamide Riboside (NR), in which Dr Brenner himself took 1,000 mg of NR and measured the effects on his blood levels of NAD+ metabolites.
The NR study was much more thorough on what they tracked. The data taken directly from that study is in the table below.
Those number were used to generate the line chart below which shows Me2PY and Me4py do not increase until after 162 minutes.
Why speed of uptake matters
Oral supplements of NR, NMN, or NAD+ go thru the digestive tract and through the portal vein into the liver.
This 2019 study by Rabinowitz shows oral NR and NMN are almost completely metabolized to NAM in the liver and then excreted in the bloodstream to the rest of the body as NAM.
As part of the salvage pathway, NAM can be converted back to NR, then NMN, and on to NAD+, but the NAM->NR is limited by availability of the required NAMPT enzyme.
Bypassing the first pass metabolism in the liver greatly increases bioavailability, and is why we created Sublingual NMN tablets and powder.
Does Slc12a8 transporter enable NMN avoid digestion to NAM ?
In research such as this 2016 study, it has long been noticed that a small quantity of NMN can reach the bloodstream in minutes.
The answer on how this happens was revealed in a study published Jan 2019 that shows the newly identified Slc12a8 protein can transport NMN to NAD+ in the small intestine, avoiding digestion to NAM.
However, this enzyme is only prevalent in the small intestine of older animals and humans.
It does not seem to be a significant amount
The study does not publish results showing HOW MUCH NMN the Slc12a8 enzyme can transport, but all previous studies such as the Rabinowitz research could not find evidence of any NAD+ at all that was created through this pathway.
According to the authors:
It is important to note that the discovery of an NMN transporter by no means diminishes the importance of uptake via dephosphorylation
There is hope Slc12a8 can be useful in future products
Future products may soon hit the market that target this pathway to help solve the bioavailability problem of NMN capsules. According to the study:
“Dr. Imai’s lab already has identified small molecules that can stimulate production of the Slc12a8 NMN transporter, applied for patents, and licensed this technology to a company in Japan”
The speedy uptake and ulilization of NMN in this first human study suggests the Slc12a8 transporter may the pathway that allows NMN to bypass digestion to NAM.
Conclusion
There were no side effects found.
There was clearly a dose dependent response, with the 500 mg dose showing more than 2x the response of a 250 mg dose.
Even the 100 mg dose showed a significant increase in NAD+ metabolites.
The speed of uptake seems to indicate some NMN is able to bypass the GI tract and liver.
