This study evaluated the efficacy of a novel formulation designed to improve absorption and efficacy of NMN.
“As awareness regarding the significance of NMN grows, recent years have witnessed an increasing exploration of innovative NMN formulations.”
Key Points:
- An NMN nano-drug delivery system using hydroxyapatite as a carrier (NMN-HAP) showed enhanced NMN absorption in mice
- NAD+ levels were higher and remained in circulation longer in mice treated with NMN-HAP
- NMN-HAP impacted distribution of NMN in tissues
- Mice administered NMN-HAP showed NMN levels in their brains that were ten times higher than those given regular NMN.
Study Evaluates Efficacy of NMN-HAP in Mice
Ninety-nine male mice were divided into four groups:
- Control group (n = 9): received standard food and water
- NMN–HAP group(n = 30): received NMN (500 mg/kg) and HAP suspension
- Free NMN group (n = 30): received NMN (500 mg/kg) suspension
- Free HAP group (n = 30): received HAP suspension
The researchers used sophisticated techniques to load NMN onto HAP to create the NMN-HAP suspension.
Blood and tissue samples were collected from the treatment groups at 1, 4, 8, 12, and 24 hours after administration of the suspensions. Samples from the control group were collected at the beginning of the study (hour 0) to establish baseline measurements.
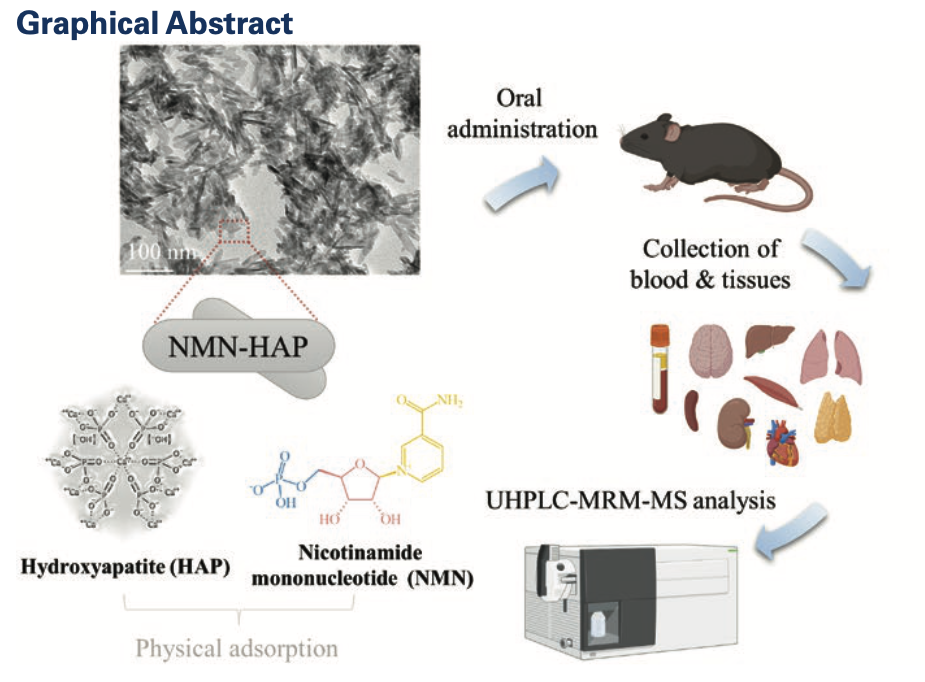
Experimental design: NMN-HAP was prepared and administered to the experimental mice. Blood and tissue samples were collected at baseline (hour 0), and hours 1, 4, 8, 12, and 24. Samples were analyzed using UHPLC-MRM-MS (an analytical technique used to identify and quantify compounds in samples).
NMN-HAP Showed Enhanced Absorption
The results showed that NMN–HAP had superior bioavailability compared to regular NMN:
- Longer to reach its highest level in the blood
- Higher peak concentration
- Lasted longer in the body
- Cleared from the body more slowly
The NMN concentrations in the plasma of mice are represented in the figure below.
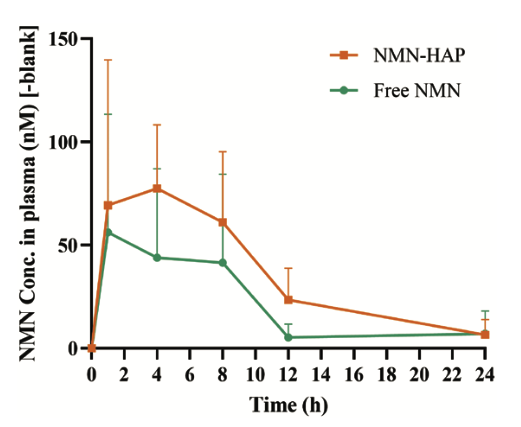
Mice that received NMN-HAP (orange bar) had higher plasma concentrations at each time point compared to mice that received regular NMN (green bar). This means that NMN-HAP is better absorbed by the body than free NMN.
“The relative bioavailability of NMN–HAP was calculated at 216.30 ± 67.93%, indicating its enhanced ability to sustain NMN release, potentially prolonging its presence in circulation and enhancing overall bioavailability.”
NMN-HAP Raised NAD+ Levels More Effectively Than Standard NMN
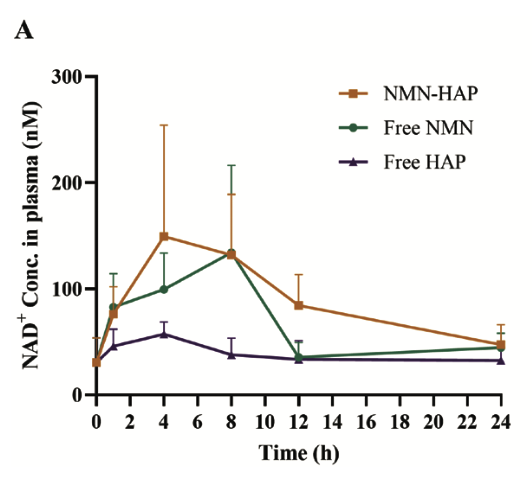
Mice given NMN-HAP (orange bar) experienced a rapid increase in NAD+ levels in the first 4 hours, followed by a gradual decline over 24 hours. Mice given regular NMN (green bar) also experienced an initial increase in NAD+ levels, but showed a more gradual increase in subsequent hours, until the 8 hour time point, where the NAD+ levels dropped significantly.
“These findings indicate a greater cumulative NAD+ accumulation in plasma after NMN–HAP administration compared with free NMN.”
NMN-HAP Showed Tissue-Specific Distribution of NMN
The researchers found that NMN-HAP impacted NMN distribution in tissues:
- Predominant accumulation in the liver
- Enhanced distribution in the brain, lungs, skeletal muscle, spleen, and thymus
- Lower concentration in the heart
NMN-HAP Increased NMN Levels in the Brain By 10-Fold
Interestingly, NMN was 10 times higher in the brain compared to regular NMN.
“These findings suggest that the utilization of the nanocarrier NMN–HAP achieves a significant improvement in brain targeting.”
These effects were also seen in NAD+ and nicotinamide riboside (NR) levels, resulting in higher accumulation in the brain and liver.
“Remarkably, NMN–HAP nanoparticles exhibited a significant enhancement in NMN delivery to two crucial tissues, the brain and liver, leading to elevated NAD+ and NR levels in these tissues.”
Conclusion
A novel delivery system effectively improved the bioavailability of NMN and led to more effective increases in NAD+ levels.
“Upon oral administration, NMN–HAP nanoparticles demonstrated prolonged circulation time and enhanced bioavailability compared with free NMN, leading to elevated plasma levels of both NAD+ and its precursor, NR.”

Hi, when was this published? I just want to be sure I am reading the most current information.
Thanks, Frank.